 
Schrödinger's equation was the first of its type, and solutions led to consequences that were very unusual and unexpected for the time. The Schrödinger equation, and its solutions, introduced a breakthrough in thinking about physics. The concept of a wavefunction is a fundamental postulate of quantum mechanics. It is not a simple algebraic equation, but in general a linear partial differential equation, describing the time-evolution of the system's wave function (also called a "state function"). 
In quantum mechanics, the analogue of Newton's law is Schrödinger's equation for a quantum system (usually atoms, molecules, and subatomic particles whether free, bound, or localized). In classical mechanics, the equation of motion is Newton's second law, ( F = m a), used to mathematically predict what the system will do at any time after the initial conditions of the system. 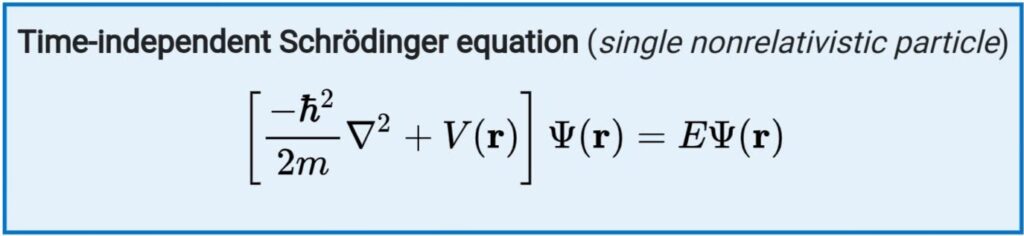
It was formulated in late 1925, and published in 1926, by the Austrian physicist Erwin Schrödinger. In quantum mechanics, the Schrödinger equation is a partial differential equation that describes how the quantum state of a physical system changes with time. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
